InnoScot Health welcomes findings of independent review into the equity of medical devices
Head of Regulatory Affairs at InnoScot Health, Elaine Gemmell NHS partner InnoScot Health has welcomed…
InnoScot Health and Heriot-Watt University bolster relationship through honorary professorship award
Elaine Gemmell, Head of Regulatory Affairs at InnoScot Health, has been conferred the title of…
BHTA calls for “urgent clarification” over how CE marking applies to medical device regulation
The Department for Business and Trade spotlight design verification and validation for medical device development…
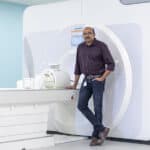
MR Coiltech partners up with InnoScot Health to win ISO accreditation for medical devices
Dr Shajan Gunamony, Director of MR Coiltech. Image: Martin Shields Glasgow-based engineering company MR CoilTech…
EU gives manufacturers more time to get certain medical devices certified under the EU MDR
The EU has taken steps to give manufacturers more time to get certain medical devices…
Government launches new strategy to boost patient access to innovative medical technology
Patients across the UK are set to benefit from access to safe, effective and innovative…
InnoScot Health celebrates 20-year anniversary with renewed focus on empowering NHS
InnoScot Health is celebrating its 20th anniversary with a renewed focus on empowering NHS Scotland…
DHSC launches call for evidence on inequalities in medical devices and technologies
An independent review by the Department of Health and Social Care (DHSC) has launched a…
Industry reacts to MHRA reports outlining the future regulation of medical devices in the UK
The Medicines and Healthcare products Regulatory Agency (MHRA) has published its response to its consultation…
Let’s Get It Clear: Understanding standards in relation to medical devices
We all come across items in our daily lives that are regulated by standards, even…